A New Hope for ALS Patients:
For the hundreds of thousands of people living with amyotrophic lateral sclerosis (ALS) worldwide, meaningful progress has been agonizingly slow. That’s what makes the latest data from NeuroSense Therapeutics so noteworthy.
The Israeli biotech company recently announced updated long-term survival results from its Phase 2b PARADIGM trial, and the numbers are hard to ignore. Patients who received PrimeC continuously throughout both the double-blind and open-label phases of the trial achieved an estimated median survival of 36.3 months, compared to just 21.4 months for those initially assigned to placebo — a difference of more than 14 months.
Perhaps even more striking is what the statistical modelling revealed. After adjusting for baseline risk factors, PrimeC treatment was associated with a 65% reduction in the risk of death compared to placebo. For a disease that typically carries a median survival of just two to five years from diagnosis, that figure carries real weight.
So what exactly is PrimeC? It’s an investigational extended-release oral formulation that combines two existing FDA-approved drugs. The trial enrolled 68 patients with ALS, and the updated survival results build on previously reported evidence of slowed disease progression and a favorable safety and tolerability profile.
Of course, important caveats remain. The trial was relatively small, and PrimeC is still an investigational therapy — it has not been approved for marketing anywhere. Phase 2 results, however promising, must be validated in larger, pivotal studies before regulators can act.
Still, NeuroSense appears encouraged. The company stated that these survival findings provide additional clinical context to support advancement of PrimeC into pivotal late-stage development, and strengthen its engagement with regulatory authorities.
For a disease community that has waited far too long for effective treatments, data like this — even at Phase 2 — is worth watching closely. The road to approval is long, but PrimeC has just taken a meaningful step forward.
This topic was covered in Great News podcast episode 36.

The Great News Podcast is your source for positive news, inspiring stories, and good news from around the world. We skip the doom and gloom of mainstream media to focus on scientific breakthroughs, environmental wins, and the inspiring news that proves the world is getting better. Join Andrew McGivern for a dose of optimism and uplifting stories that will change your perspective on human progress.
It is easy to find the
Keep looking for the good in the world, because it is not only there – its everywhere.
This episode is brought to you by the Daily Quote. The podcast designed to kickstart your day in a positive way.
Today, we are diving into some truly groundbreaking advancements in medicine, biotechnology, and sustainable energy. What if a gene editor could replicate and spread like a virus to ensure it reaches every cell that needs a cure? That is our lead story today.
Plus, we have a ”rechargeable sun battery” that outperforms lithium-ion, tiny bubbles that shatter cancer’s defenses, and a city-wide success story in wiping out Dengue fever.
CRISPR Gene Editor Replicates and Spreads Like a Virus
New Sun Battery Beats Lithium-Ion
Bubbles and Ultrasound Break Soften Tumour Walls for Better Treatment
And don’t forget to stick around for the speed round, where we’ll dive into even more great news.Prime C Shows Striking Survival Benefit in ALS Clinical Trial
Inhalable Treatment Could Replace Months of Pills for TB
Could a “Longevity Protein” Slow How We Age?An Inflammation Suppressor Decreases Mortality
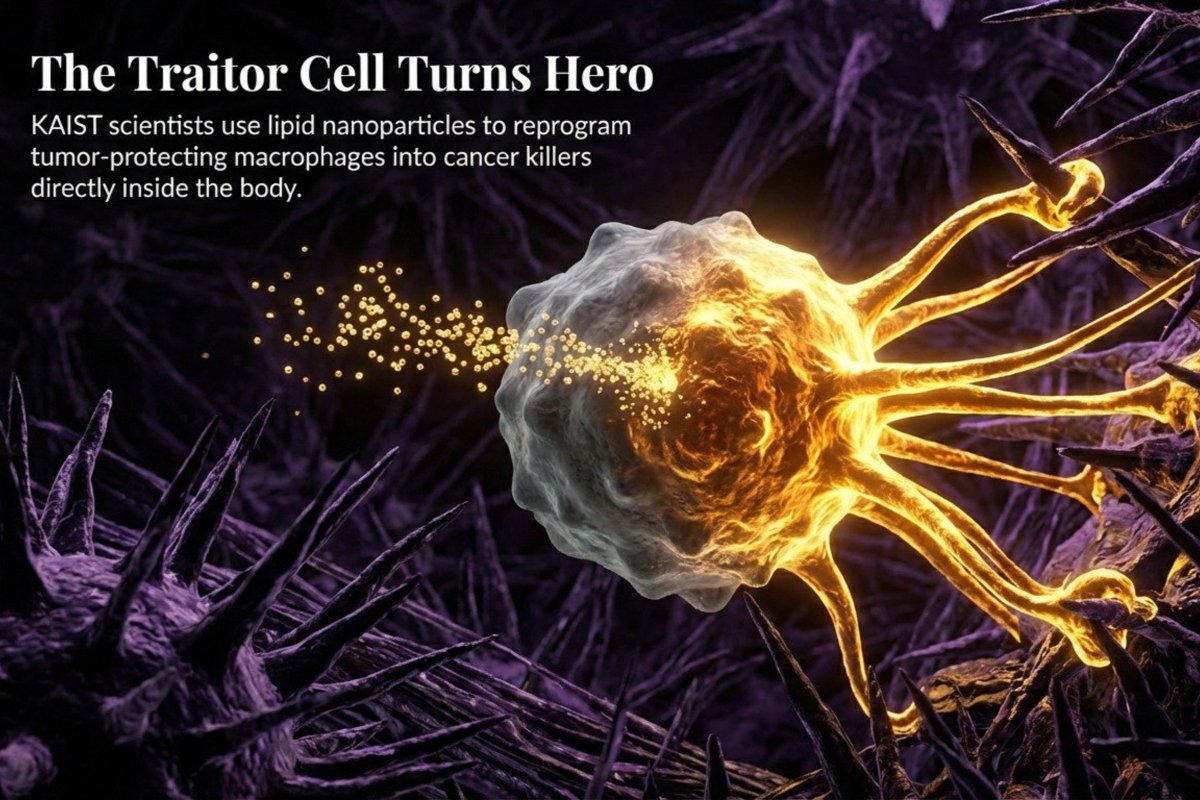
Turning Our Own DNA Against Cancer
Until next time, keep looking for the good in the world, because it’s not only there—it’s everywhere.

Source: Longevity Technology