Cracking the “Undruggable” Code:
For decades, certain cancer-driving proteins have been considered essentially off-limits to drug developers. These proteins, called transcription factors, act like master switches in our cells, controlling which genes turn on and off. In cancer, they get stuck in the “on” position, fueling uncontrolled growth. The problem? Their smooth, featureless surfaces give conventional drugs almost nothing to grip onto.
Now, researchers at the University of Bath may have found an elegant way around this long-standing obstacle.
The clever trick: staple them inside bacteria
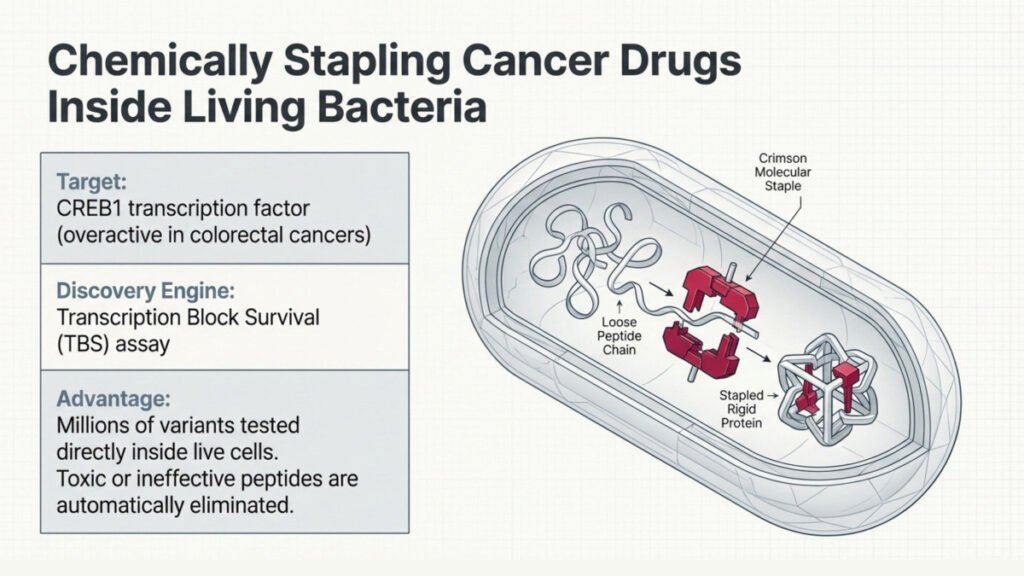
The Bath team has developed a technology that uses bacteria to build, chemically stabilize, and test millions of potential drug molecules inside living cells, all in one go. The molecules in question are peptides, short chains of amino acids that can be designed to interfere with specific proteins.
The key innovation is the “stapling” step. Each bacterium produces a different peptide, which is then chemically modified inside the living cell, acting like a molecular staple, locking the peptide into a defined shape it would not normally adopt. This rigid shape is what makes the peptide stable and effective enough to work inside a cell.
What makes the approach genuinely clever is that the chemistry happens while the drug is being tested. Only bacteria that produce peptides which are both effective and non-toxic survive, meaning the biological process itself filters out the failures and surfaces the winners naturally.
Faster, greener, and scalable
Beyond its scientific ingenuity, the method also sidesteps many of the practical headaches of conventional drug discovery. Traditional methods require peptides to be made, purified, chemically modified, and purified again — whereas stapled peptides here can be recovered directly from the cell in a single, simplified step. The process also avoids the toxic solvents typically required, making it cleaner and more cost-effective.
A real target, real results
To put the technology through its paces, the team used it to go after CREB1, a transcription factor that is overactive in a broad range of cancers, including colorectal cancer. The peptides they identified didn’t just bind to the target in theory — they were shown to enter human cancer cells grown in the lab, shut down CREB1-controlled pathways, and selectively kill cancer cells.
That last word, selectively, is crucial. One of the biggest challenges in cancer treatment is sparing healthy cells from the damage. Peptides that can distinguish between normal and cancerous behavior represent a meaningful step forward.
What comes next
The research, published in Cell Chemical Biology, is still in its early stages. The next hurdle is demonstrating these results hold up in more complex tissue models and animal studies. But the platform itself is the real headline: a system that lets biology and chemistry work in concert to rapidly discover a new class of drugs for cancers that have long resisted treatment.
If it translates, it could open the door to tackling not just CREB1 but a whole family of transcription factors that scientists have been eyeing (and struggling to reach) for years.
This topic is featured in Great News podcast episode 39.

The Great News Podcast is your source for positive news, inspiring stories, and good news from around the world. We skip the doom and gloom of mainstream media to focus on scientific breakthroughs, environmental wins, and the inspiring news that proves the world is getting better. Join Andrew McGivern for a dose of optimism and uplifting stories that will change your perspective on human progress.
It is easy to find the
Keep looking for the good in the world, because it is not only there – its everywhere.
The Great News podcast is brought to you by the Daily Quote.
Today, a discovery that could turn our oceans into a source of clean, green fuel. That's our lead story today.
Plus, we’ll look at a ”molecular staple” for hard-to-treat cancers, a way to silence cholesterol without permanently altering your DNA, and a wireless system that’s twice as efficient as Wi-Fi.
Turning Sunlight and Seawater into Hydrogen
How Stapled Peptides Could Change Cancer Treatment
How Laser Tech Is Rewriting the Rules of WirelessAnd don’t forget to stick around for the speed round where we’ll celebrate a historic victory against an ancient disease.Chile Eliminates Leprosy
Revolutionizing Brain Research and Treatment!
How CAR T Therapy Is Learning to Fight Cancer Smarter
If you like the Great News Podcast, you will love the Great News Letter. Because the Great News Podcast is Great but the Great News Letter is Greater!
The Great News Letter is an email newsletter that you can subscribe to for FREE and get all the good news delivered to your email inbox.
For this episode we got some feedback from Brian de V. You can leave feedback for the show too by going to greatnewspodcast.com/feedback.
Or if you listen in Spotify – leave a comment right there as you listen.
Until next time… and there will be a next time.
Keep looking for the good in the world because no only is it there, its everywhere!

Source: