Liquid Gallium to the Rescue!
Hey clean energy enthusiasts! Imagine a world where we can produce hydrogen fuel directly from sunlight and even seawater, without needing fancy purified water or tons of electricity. Sounds like science fiction, right? Well, researchers at the University of Sydney are making it a reality with a groundbreaking method using – get this – liquid gallium!
Hydrogen is a super promising clean fuel, but making it at scale has been a challenge due to cost and the need for pure water. But this new technique changes the game!
At the heart of this innovation is gallium, a metal that melts just above room temperature. When these liquid gallium particles are mixed with water (yes, even seawater!) and exposed to light, a magical reaction happens: hydrogen is released! The gallium reacts with the water to form gallium oxyhydroxide, freeing up those precious hydrogen molecules.
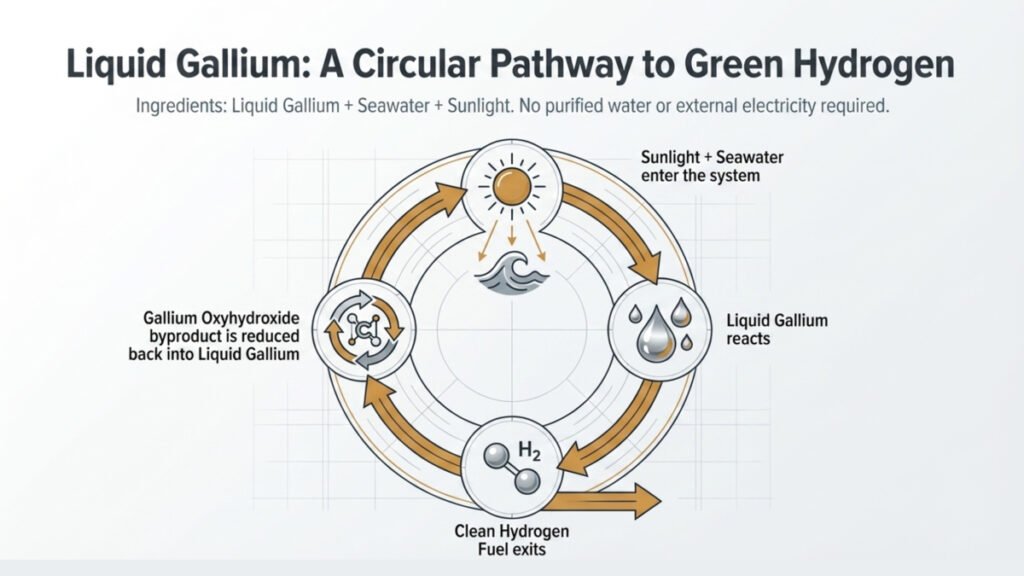
What’s really cool is that this process doesn’t need external electricity or complex infrastructure. Just sunlight (or artificial light) and water! This means we could potentially produce hydrogen in coastal areas or regions with limited freshwater resources, making clean fuel more accessible globally.
And it gets even better: the process is circular! After the hydrogen is extracted, the gallium oxyhydroxide can be converted back into gallium and reused for more hydrogen production. Talk about sustainable!
While the team is still working on increasing efficiency for commercial use (they’ve hit 12.9% so far, which is pretty competitive!), this discovery opens up exciting possibilities for a future powered by clean, green hydrogen. It’s a simple yet brilliant observation that could have a massive impact on our energy landscape. Stay tuned for more on this incredible innovation!
This topic is featured in Great News podcast episode 39.

The Great News Podcast is your source for positive news, inspiring stories, and good news from around the world. We skip the doom and gloom of mainstream media to focus on scientific breakthroughs, environmental wins, and the inspiring news that proves the world is getting better. Join Andrew McGivern for a dose of optimism and uplifting stories that will change your perspective on human progress.
It is easy to find the
Keep looking for the good in the world, because it is not only there – its everywhere.
The Great News podcast is brought to you by the Daily Quote.
Today, a discovery that could turn our oceans into a source of clean, green fuel. That's our lead story today.
Plus, we’ll look at a ”molecular staple” for hard-to-treat cancers, a way to silence cholesterol without permanently altering your DNA, and a wireless system that’s twice as efficient as Wi-Fi.
Turning Sunlight and Seawater into Hydrogen
How Stapled Peptides Could Change Cancer Treatment
How Laser Tech Is Rewriting the Rules of WirelessAnd don’t forget to stick around for the speed round where we’ll celebrate a historic victory against an ancient disease.Chile Eliminates Leprosy
Revolutionizing Brain Research and Treatment!
How CAR T Therapy Is Learning to Fight Cancer Smarter
If you like the Great News Podcast, you will love the Great News Letter. Because the Great News Podcast is Great but the Great News Letter is Greater!
The Great News Letter is an email newsletter that you can subscribe to for FREE and get all the good news delivered to your email inbox.
For this episode we got some feedback from Brian de V. You can leave feedback for the show too by going to greatnewspodcast.com/feedback.
Or if you listen in Spotify – leave a comment right there as you listen.
Until next time… and there will be a next time.
Keep looking for the good in the world because no only is it there, its everywhere!

Source: