A Surprising New Frontier in Cancer Treatment
What if the next breakthrough in cancer therapy came not from a drug, but from a living organism — one found in ordinary soil?
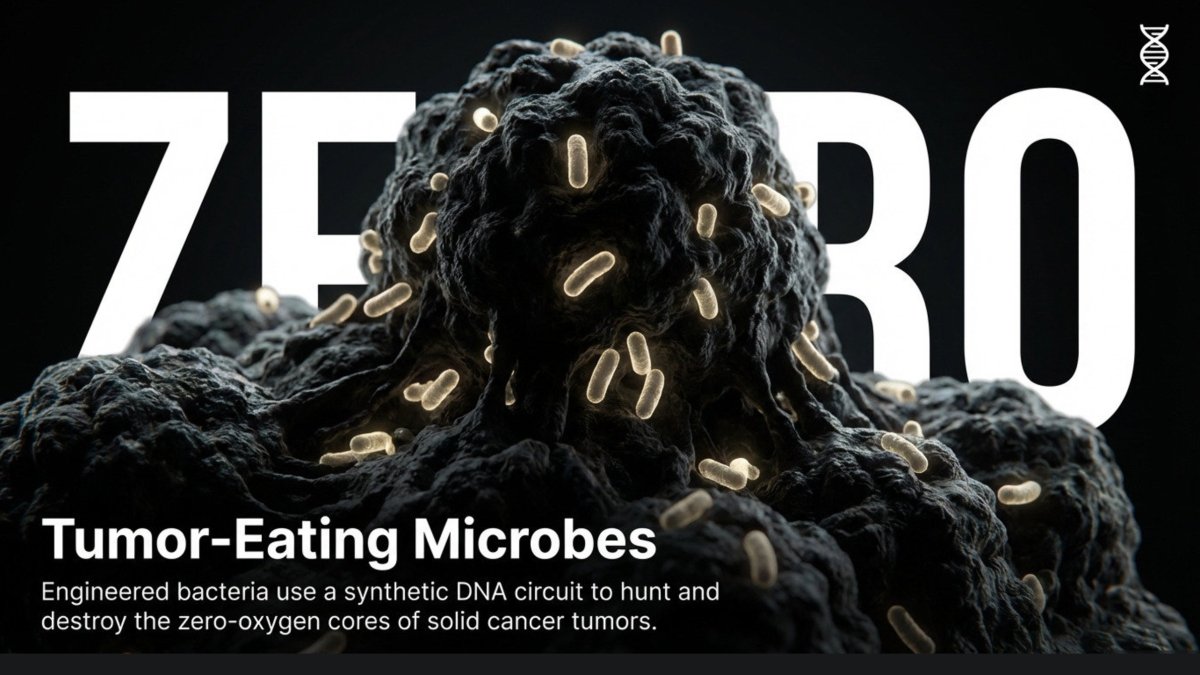
Researchers at the University of Waterloo are betting on exactly that. Their team is engineering a bacterium called Clostridium sporogenes to seek out and destroy solid tumors from the inside out, in a genuinely novel approach to one of medicine’s oldest challenges.
The Biology Behind the Idea
The concept exploits a quirk of tumor biology. At the core of most solid tumors, there is a region of dead cells that is completely devoid of oxygen. For C. sporogenes — an anaerobic organism that thrives in oxygen-free environments — this is an ideal habitat. Introduce its spores into the body, and they migrate naturally to these tumor cores, begin feeding on the available nutrients, and multiply.
“Bacteria spores enter the tumor, finding an environment where there are lots of nutrients and no oxygen, which this organism prefers,” explained Dr. Marc Aucoin, a chemical engineering professor at Waterloo, “and so it starts eating those nutrients and growing in size.”
In principle, the bacteria colonize the tumor’s necrotic core and begin consuming it from within.
The Problem — And the Clever Fix
There is a catch. When the bacteria reach the outer edges of a tumor — where living cancer cells still reside — they encounter trace amounts of oxygen. This kills them before they can finish the job.
To solve this, the Waterloo team took a two-step engineering approach. First, they introduced a gene from a related, more oxygen-tolerant bacterium, allowing C. sporogenes to survive longer near the tumor’s periphery. Then they tackled the obvious safety concern: how do you prevent oxygen-resistant bacteria from growing somewhere dangerous, like the bloodstream?
Their answer was quorum sensing — a natural phenomenon in which bacteria communicate via chemical signals. Only when enough bacteria have accumulated within a tumor does the signal become strong enough to switch on the oxygen-resistance gene. It’s a built-in timer that activates the capability only when, and where, it’s needed.
Dr. Brian Ingalls, a professor of applied mathematics at Waterloo, described the mechanism in elegant terms: “Using synthetic biology, we built something like an electrical circuit, but instead of wires we used pieces of DNA. Each piece has its job. When assembled correctly, they form a system that works in a predictable way.”
Where Things Stand
So far, the researchers have demonstrated each component separately — oxygen tolerance in one study, and the quorum sensing circuit (validated using a green fluorescent protein marker) in another. The next step is to combine both modifications into a single bacterium and test it against a tumor in pre-clinical trials.
The project grew from the doctoral work of Bahram Zargar, now co-founder of CREM Co Labs, a Toronto-based microbiology company that partnered on the research. Former Waterloo doctoral student Dr. Sara Sadr played a leading role in the lab work.
Why This Matters
Solid tumors — including many pancreatic, colorectal, and lung cancers — remain notoriously difficult to treat. Their dense structure limits drug penetration, and their oxygen-deprived cores are unreachable by immune cells that require oxygen to survive. That same inhospitable interior, long considered a liability, is precisely what this approach turns into an asset.
The research is still early-stage, and a long path to the clinic remains. But the underlying logic is sound, the engineering is inventive, and the potential — a living, self-targeting therapeutic that navigates to where it’s needed most — is hard to ignore.
Source: University of Waterloo / ACS Synthetic Biology
This topic was featured in Great News podcast episode 35.

The Great News Podcast is your source for positive news, inspiring stories, and good news from around the world. We skip the doom and gloom of mainstream media to focus on scientific breakthroughs, environmental wins, and the inspiring news that proves the world is getting better. Join Andrew McGivern for a dose of optimism and uplifting stories that will change your perspective on human progress.
It is easy to find the
Keep looking for the good in the world, because it is not only there – its everywhere.
This podcast is brought to you by the Daily Quote. The podcast designed to kickstart your day in a positive way!
Today, we are exploring a medical breakthrough that uses ”hungry” bacteria to fight cancer, a massive electric flying taxi taking to the skies in China, and a successful harvest of chickpeas grown in actual moon dirt
Plus, we’ll look at a new computer chip that gives self-driving cars reflexes four times faster than the human brain
Bacteria that eats cancer from the inside out
Robot Cars react 4X faster than humans
And stick around for our speed round, where we’ll cover even more great news!
US Forests fighting harder against climate change
Gold Supraballs capture 90% of solar spectrum
A few changes at home reduce asthma attacks
New additive increases organic solar cell efficiency
If you like the Great News Podcast, You'll love the great news letter. Because the Great News Podcast is Great! But the Great News Letter is Greater!
The Great News Letter is FREE and includes all the good news stories.
Source: Medical Life Sciences News