Antibiotic resistance is one of the most serious and underreported threats to global public health. Bacteria are evolving faster than our ability to develop new drugs to counter them, and the consequences are already being felt in hospitals, care homes, and beyond. Without urgent intervention, estimates suggest antibiotic-resistant infections could be responsible for more than 10 million deaths per year worldwide by 2050 — surpassing cancer as a leading cause of death.
Most current approaches to this problem focus on slowing resistance or working around it. A new breakthrough from the University of California San Diego is doing something far more ambitious: actively reversing it.
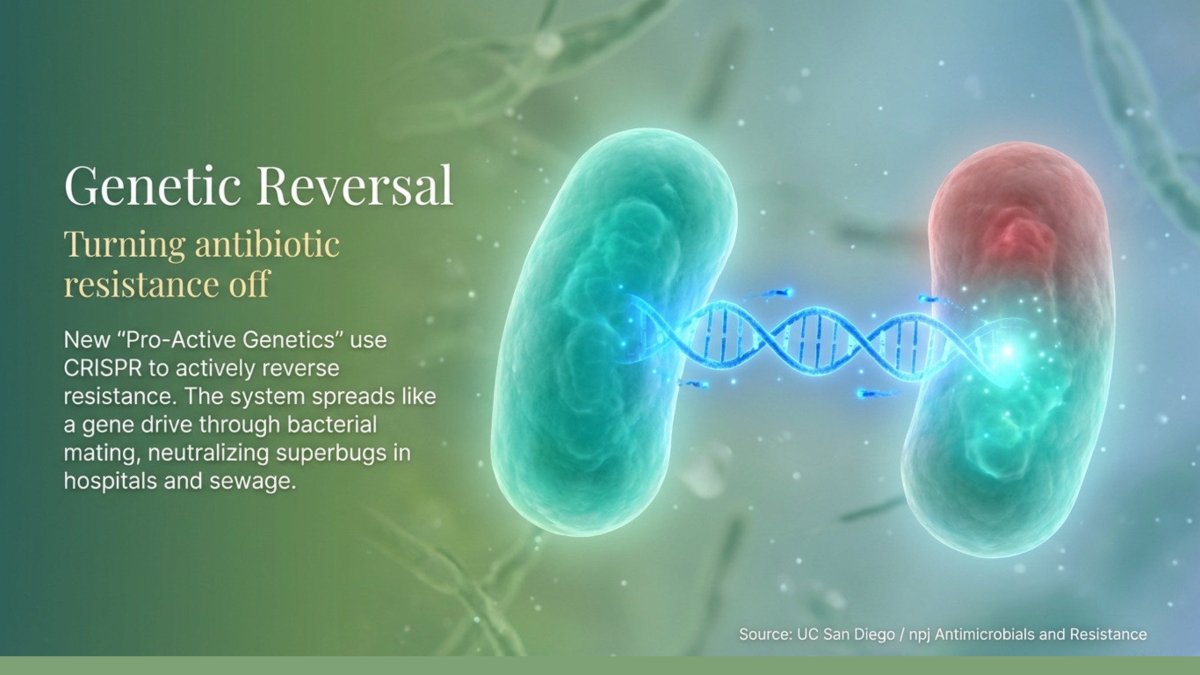
Researchers from UC San Diego’s School of Biological Sciences have developed a next-generation CRISPR-based tool called pPro-MobV, which is designed to strip antibiotic resistance from bacterial populations rather than simply trying to outmanoeuvre it. The concept draws inspiration from gene drive technology — a technique already being used in insects to disrupt the spread of malaria — and applies it to bacteria for the first time.
The system works by introducing a genetic cassette that targets and disables antibiotic-resistant elements carried on plasmids — circular strands of DNA that replicate within bacterial cells and are one of the primary mechanisms through which resistance spreads. Crucially, the new version of this technology spreads the disabling genetic elements through a process called conjugal transfer — essentially bacterial mating — allowing the cassette to propagate through a population naturally, using the bacteria’s own biological machinery against them.
One of the most significant aspects of the research is that the team demonstrated it working inside bacterial biofilms. Biofilms are communities of microorganisms that contaminate surfaces and contribute to the spread of serious disease; they are particularly difficult to treat because their layered structure limits the ability of antibiotics to penetrate through. The fact that this technology can operate within biofilms opens up potential applications not just in clinical settings, but in aquaculture, sewage treatment plants, and environmental remediation — places where antibiotic-resistant bacteria flourish and from which they spread.
Professor Justin Meyer, who studies the evolutionary adaptations of bacteria and viruses, noted that this is one of the very few approaches he is aware of that can actively reverse the spread of antibiotic-resistant genes, rather than simply slowing or coping with their spread. That distinction matters enormously. The difference between managing a problem and reversing it is the difference between a holding action and a genuine solution.
The research, published in the Nature journal npj Antimicrobials and Resistance, also found that the system’s components could be delivered by bacteriophage — viruses that are natural predators of bacteria — opening up another avenue for targeted deployment. And a safety mechanism has been built in that allows the genetic cassette to be removed if needed.
There is still a long road from laboratory success to clinical application, and the researchers are clear that this technology needs further development and testing. But in a field where the news has largely been grim for decades, a tool that can actively roll back resistance rather than simply delay it represents a genuinely new direction — and a reason for cautious optimism.
The bacteria may be evolving. But so are we.
This topic was covered in Great News podcast episode 31.

The Great News Podcast is your source for positive news, inspiring stories, and good news from around the world. We skip the doom and gloom of mainstream media to focus on scientific breakthroughs, environmental wins, and the inspiring news that proves the world is getting better. Join Andrew McGivern for a dose of optimism and uplifting stories that will change your perspective on human progress.
It is easy to find the
Keep looking for the good in the world, because it is not only there – its everywhere.
Today we’re diving into some truly groundbreaking advancements in biotechnology, sustainable energy, and global economics. What if we could actually reverse antibiotic resistance in bacteria instead of just trying to keep up with it?
Plus, we have packaging that grows itself from mushrooms, a giga-scale geothermal breakthrough in Utah, and new data showing the world is more equal than you might think.
- What If We Could Teach Bacteria to Forget How to Resist Antibiotics?
- Global Inequality Declines Substantially
- Geothermal Breakthrough
And don't forget to stick around for the speed round where we'll dive into even more great news.
- The Screen Time Panic May Be Missing the Point
- China grants commercial flying taxi licenses
- The Future of Hair Loss Treatment Might Just Be a Hat
- Your Immune System Could Be the Key to Slowing Ageing
- AI has identified 15 new magnetic materials
And don't forget to follow the Daily Quote

Source: News Medical Life Sciences